Fe3+ Scn Fescn2+ Equilibrium Constant
This experiment outlines the techniques necessary to determine the equilibrium abiding for the formation of an fe(III) thiocyanate circuitous ion (FeSCNii+) from Fe3+ and SCN- . The quantitative preparation of several solutions and subsequent measurement of the solution absorbance using a spectrophotometer are the techniques that will be used in this experiment. The absorbance measurement gives the concentration of FeSCN2+. The concentrations of Atomic number 26iii+ and SCN- are obtained as the departure between the initial concentration and the concentration consumed past the germination of the FeSCN2+. The combined concentrations will be used to calculate an equilibrium constant for the formation of the complex.
The reaction for the formation of the dark red FeSCN2+ complex ion is very simple:
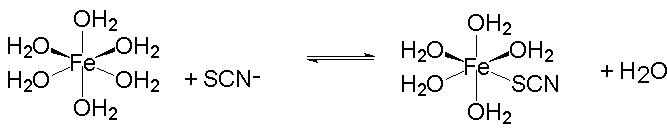
It has an equilibrium constant, Thou, given by:
Starting with known amounts of iron(III) and thiocyanate, and measuring the amount of FeSCN2+ ion formed at equilibrium, one can calculate the equilibrium amounts of iron(Iii) and thiocyanate ions. From a knowledge of the equilibrium amounts of all 3 ions, the equilibrium constant for the reaction may be calculated.
2. Experimental Process.
The handling of the instrument volition be demonstrated.
Ideally the five equilibrium constants obtained from the five sets of data should exist the same. How closely they really match will be a function of the intendance you lot have in carrying the experiment.
Information technology is very of import that you lot empathise what you lot are doing at all times. Proper labeling should help.
A. Standard Solution.
The instrument must be calibrated by a solution in which the concentration of the complex ion is known. This is achieved by preparing i solution where the starting concentration of iron(Iii) ions exceeds the starting concentration of thiocyanate ions past two orders of magnitude. Under these conditions, of backlog atomic number 26, it may be safe assumed that all the original thiocyanate volition take been used to form the complex ion (LeChâtelier'southward principle). Thus the concentration of the circuitous may exist assumed to be equal to the initial concentration of thiocyanate. The concentration of the circuitous in all the other solutions is and so adamant by the instrument as a fraction of the concentration of the complex in this, the standard solution.
Pipet 5.0 mL of 0.10 Thou iron(Iii) nitrate into each of five 150 mm exam tube. Add the post-obit amounts of KSCN and diluted nitric acid to each of the tubes:
#1 0 mL KSCN and 5 mL nitric acid
#2 0.2 mL KSCN and 4.8 mL nitric acid
#3 0.four mL KSCN and 4.6 mL nitric acid
#iv 0.half-dozen mL KSCN and 4.4 mL nitric acid
#5 0.8 mL KSCN and 4.6 mL nitric acid
Mix them well. Characterization information technology. This is your calibration set of solutions. It is assumed that the concentration of the FeSCN2+ complex in this solution is exactly equal to full concentration of SCN.
Using the EXCEL program, plot the Absorbance (A) equally a role of thiocyanate concentration; this is your calibration bend. The trend line should be a straight line with the slope of e and intercept b
A=e C+b
You volition apply the value of east in farther calculations.
B. Working Solutions.
Label five 150 mm exam tubes from 1 to 5. Pipet 5.0 mL of 2.0 mM Atomic number 26iii+ into each.
Add the following amounts of KSCN and diluted nitric acid to each of the tubes:
#1 0.5 mL KSCN and four.five mL nitric acid
#2 1 mL KSCN and iv mL nitric acrid
#3 2 mL KSCN and 3 mL nitric acid
#4 3 mL KSCN and 2 mL nitric acrid
#five 4 mL KSCN and 1 mL nitric acid
Full volume in each tube is 10 ml (bank check it!).
Measure absorbance of each solution.
C. Determination of Absorbance
The Spectronic xx spectrophotometer will be used to measure the amount of light existence absorbed at 450 nm, the wavelength at which the thiocyanatoiron(3) circuitous absorbs visible light. Musical instrument controls will exist demonstrated by your teacher. Connect the instrument to a 115 V Ac outlet, and let it warm-up for ten-15 minutes.
.

The instrument must exist calibrated. Ready the wavelength to 450 nm with the WAVELENGTH control.
With nothing in the CELL COMPARTMENT, use the Nighttime Electric current control (the aforementioned control that turns the instrument on and off) to set the musical instrument to read 0% Transmittance (black scale).


Fill a cuvet with deionized water, and dry the outside and wipe it clean of fingerprints with Kimwipe. Insert the examination tube into the Prison cell COMPARTMENT every bit far equally it will go. Set the instrument to read 100% Transmittance with the LIGHT control. The musical instrument is now calibrated. The settings of the controls must not exist changed from now on, or you will have to recalibrate.
Make full another cuvet with your solution.. Wipe the outside with tissue and and so insert it into the CELL COMPARTMENT (later on removing the test tube containing the deionized water, of form). Decide the absorbance and record it.
CALCULATIONS
Calculate initial concentrations of atomic number 26 and of thiocyanate in each of your 5 solutions.
Utilise your calibration to determine the concentration of FeSCN2+
[ FeSCN2+]= A/ eastward
Decrease the [ FeSCN2+] from the initial concentration of iron: this is your concentration of Fe3+ at equilibrium.
Decrease the [ FeSCN2+] from the initial concentration of thiocyanate: this is your concentration of SCN- at equilibrium.
Put the concentrations you lot accept calculated in equation
Chemical Equilibrium:
Determination of an Equilibrium Constant of a Complex.
Iii. Data/Report.
Calibration plot:
To summate the concentration of KSCN, use proportion:
Concentration KSCN = (Standard concentration) x (Volume KSCN) / (Total volume)
Your standard concentration is ii.0 mM = 2.0x10-three M
Total volume is 10 mL (check it).
| Number | Volume KSCN | Concentration KSCN | Absorbance |
| 1 | 0 | 0 | 0. |
| 2 | 0.2 | ,M | 0. |
| 3 | 0.4 | ,M | 0. |
| 4 | 0.vi | ,Yard | 0. |
| 5 | 0.8 | ,M | _. |
The slope of the calibration line is ___________
This is tooth absorptivity of FeSCNii+ ion.
To summate the initial concentration of SCN, utilise proportion:
Initial SCN concentration = (Standard concentration) x (Volume KSCN) / (Total volume)
Your standard concentration is 2.0 mM = ii.0x10-three M
Total book is 10 mL (check information technology).
To calculate the initial concentration of iron, apply proportion:
Initial Atomic number 26 concentration = (Standard concentration) ten (Volume Fe) / (Total volume)
Your standard concentration is 2.0 mM = two.0x10-3 M.
[ FeSCNii+]= A/ east
| No. | 5Fe | VSCN | Initial Fe concentr. | Initial SCN concentr. | Absorb. | [FeSCN2+] | [Atomic number 263+] | [SCN-] | Mc (in Chiliad -i ) |
| ane | 5.0 | 0.5 | ,Chiliad | ,G | . | ,M | ,Chiliad | ,M | . |
| 2 | 5.0 | one | ,G | ,M | . | ,K | ,M | ,M | . |
| three | five.0 | 2 | ,M | ,M | . | ,M | ,M | ,M | . |
| iv | 5.0 | iii | ,M | ,Yard | . | ,M | ,G | ,Yard | . |
| v | 5.0 | 4 | ,M | ,M | . | ,M | ,M | ,Grand | . |
| Boilerplate: | _._x10 |
Fe3+ Scn Fescn2+ Equilibrium Constant,
Source: https://staff.buffalostate.edu/nazareay/che112/ex6.htm
Posted by: berniertheirm.blogspot.com


0 Response to "Fe3+ Scn Fescn2+ Equilibrium Constant"
Post a Comment