Chemical Formula For Aluminum Fluoride
| |||
| Names | |||
|---|---|---|---|
| IUPAC name Fluoride[one] | |||
| Identifiers | |||
| CAS Number |
| ||
| 3D model (JSmol) |
| ||
| ChEBI |
| ||
| ChEMBL |
| ||
| ChemSpider |
| ||
| Gmelin Reference | 14905 | ||
| KEGG |
| ||
| MeSH | Fluoride | ||
| PubChem CID |
| ||
| UNII |
| ||
| InChI
| |||
| SMILES
| |||
| Properties | |||
| Chemical formula | F − | ||
| Tooth mass | 18.998403 163 g·mol−1 | ||
| Conjugate acrid | Hydrogen fluoride | ||
| Thermochemistry | |||
| Std molar | 145.58 J/mol Chiliad (gaseous)[2] | ||
| Std enthalpy of | −333 kJ mol−1 | ||
| Related compounds | |||
| Other anions |
| ||
| Except where otherwise noted, data are given for materials in their standard country (at 25 °C [77 °F], 100 kPa). Infobox references | |||
Fluoride ()[3] is an inorganic, monatomic anion of fluorine, with the chemical formula F −
(also written [F] −
), whose salts are typically white or colorless. Fluoride salts typically have distinctive biting tastes, and are odorless. Its salts and minerals are important chemical reagents and industrial chemicals, mainly used in the production of hydrogen fluoride for fluorocarbons. Fluoride is classified as a weak base of operations since it only partially associates in solution, only full-bodied fluoride is corrosive and can attack the pare.
Fluoride is the simplest fluorine anion. In terms of charge and size, the fluoride ion resembles the hydroxide ion. Fluoride ions occur on Earth in several minerals, especially fluorite, but are nowadays merely in trace quantities in bodies of h2o in nature.
Classification [edit]
Fluorides include compounds that incorporate ionic fluoride and those in which fluoride does not dissociate. The nomenclature does not distinguish these situations. For case, sulfur hexafluoride and carbon tetrafluoride are not sources of fluoride ions nether ordinary conditions.
The systematic name fluoride, the valid IUPAC name, is determined according to the additive nomenclature. However, the name fluoride is as well used in compositional IUPAC nomenclature which does not accept the nature of bonding involved into account. Fluoride is too used not-systematically, to draw compounds which release fluoride upon dissolving. Hydrogen fluoride is itself an example of a non-systematic name of this nature. However, it is likewise a trivial proper name, and the preferred IUPAC name for fluorane.[ citation needed ]
Occurrence [edit]

Fluorine is estimated to be the 13th-most arable element in Earth's crust and is widely dispersed in nature, entirely in the form of fluorides. The vast bulk is held in mineral deposits, the almost commercially important of which is fluorite (CaF2).[4] Natural weathering of some kinds of rocks, as well equally human activities, releases fluorides into the biosphere through what is sometimes called the fluorine cycle.
- In h2o
Fluoride is naturally present in groundwater, fresh and saltwater sources, likewise as in rainwater, especially in urban areas.[5] Seawater fluoride levels are usually in the range of 0.86 to i.4 mg/L, and average 1.1 mg/L[six] (milligrams per litre). For comparison, chloride concentration in seawater is about 19 g/L. The low concentration of fluoride reflects the insolubility of the alkaline earth fluorides, e.g., CaF2.
Concentrations in fresh water vary more significantly. Surface water such as rivers or lakes generally contains betwixt 0.01–0.3 mg/L.[7] Groundwater (well water) concentrations vary even more, depending on the presence of local fluoride-containing minerals. For example, natural levels of under 0.05 mg/L have been detected in parts of Canada only upwards to 8 mg/50 in parts of China; in general levels rarely exceed 10 mg/litre[8]
- In parts of Asia the groundwater can contain dangerously high levels of fluoride, leading to serious health problems.[9]
- Worldwide, 50 one thousand thousand people receive water from water supplies that naturally have shut to the "optimal level".[10]
- In other locations the level of fluoride is very low, sometimes leading to fluoridation of public h2o supplies to bring the level to effectually 0.vii–1.two ppm.
- Mining can increment local fluoride levels[11]
Fluoride tin can be present in rain, with its concentration increasing significantly upon exposure to volcanic activity[12] or atmospheric pollution derived from burning fossil fuels or other sorts of industry,[xiii] [14] particularly aluminium smelters.[xv]
- In plants
All vegetation contains some fluoride, which is absorbed from soil and water.[eight] Some plants concentrate fluoride from their environment more than others. All tea leaves contain fluoride; yet, mature leaves contain as much as 10 to xx times the fluoride levels of young leaves from the same plant.[16] [17] [eighteen]
Chemical properties [edit]
Basicity [edit]
Fluoride can act every bit a base of operations. Information technology tin can combine with a proton (H+):
This neutralization reaction forms hydrogen fluoride (HF), the conjugate acid of fluoride.
In aqueous solution, fluoride has a pK b value of 10.eight. It is therefore a weak base, and tends to remain equally the fluoride ion rather than generating a substantial amount of hydrogen fluoride. That is, the following equilibrium favours the left-hand side in water:
-
F− + H2O HF + HO−
(ii)
However, upon prolonged contact with moisture, soluble fluoride salts will decompose to their respective hydroxides or oxides, as the hydrogen fluoride escapes. Fluoride is distinct in this regard amongst the halides. The identity of the solvent can have a dramatic upshot on the equilibrium shifting it to the correct-hand side, greatly increasing the rate of decomposition.
Structure of fluoride salts [edit]
Salts containing fluoride are numerous and adopt myriad structures. Typically the fluoride anion is surrounded by 4 or vi cations, as is typical for other halides. Sodium fluoride and sodium chloride adopt the same structure. For compounds containing more than one fluoride per cation, the structures often deviate from those of the chlorides, as illustrated by the main fluoride mineral fluorite (CaF2) where the Catwo+ ions are surrounded by eight F− centers. In CaCl2, each Catwo+ ion is surrounded by 6 Cl− centers. The difluorides of the transition metals often adopt the rutile structure whereas the dichlorides have cadmium chloride structures.
Inorganic chemistry [edit]
Upon treatment with a standard acrid, fluoride salts convert to hydrogen fluoride and metal salts. With strong acids, it can be doubly protonated to give H
2 F +
. Oxidation of fluoride gives fluorine. Solutions of inorganic fluorides in water comprise F− and bifluoride HF −
2 .[19] Few inorganic fluorides are soluble in water without undergoing significant hydrolysis. In terms of its reactivity, fluoride differs significantly from chloride and other halides, and is more strongly solvated in protic solvents due to its smaller radius/charge ratio. Its closest chemical relative is hydroxide, since both have similar geometries.
Naked fluoride [edit]
Most fluoride salts deliquesce to give the bifluoride (HF −
two ) anion. Sources of true F− anions are rare considering the highly basic fluoride anion abstracts protons from many, fifty-fifty adventitious, sources. Relative unsolvated fluoride, which does exist in aprotic solvents, is called "naked". Naked fluoride is a strong Lewis base of operations,[20] and a powerful nucleophile. Some quaternary ammonium salts of naked fluoride include tetramethylammonium fluoride and tetrabutylammonium fluoride.[21] Cobaltocenium fluoride is another instance.[22] Withal, they all lack structural characterization in aprotic solvents. Because of their high basicity, many and then-called naked fluoride sources are in fact bifluoride salts. In late 2016 imidazolium fluoride was synthesized that is the closest approximation of a thermodynamically stable and structurally characterized example of a "naked" fluoride source in an aprotic solvent (acetonitrile).[23] The sterically demanding imidazolium cation stabilizes the detached anions and protects them from polymerization.[24] [25]
Biochemistry [edit]
At physiological pHs, hydrogen fluoride is usually fully ionised to fluoride. In biochemistry, fluoride and hydrogen fluoride are equivalent. Fluorine, in the form of fluoride, is considered to be a micronutrient for man health, necessary to preclude dental cavities, and to promote salubrious os growth.[26] The tea institute (Camellia sinensis Fifty.) is a known accumulator of fluorine compounds, released upon forming infusions such as the common beverage. The fluorine compounds decompose into products including fluoride ions. Fluoride is the most bioavailable form of fluorine, and every bit such, tea is potentially a vehicle for fluoride dosing.[27] Approximately, 50% of captivated fluoride is excreted renally with a xx-four-hour period. The remainder tin can be retained in the oral cavity, and lower digestive tract. Fasting dramatically increases the rate of fluoride absorption to near 100%, from a 60% to 80% when taken with nutrient.[27] Per a 2013 report, it was plant that consumption of one litre of tea a day, tin can potentially supply the daily recommended intake of four mg per day. Some lower quality brands can supply up to a 120% of this amount. Fasting can increment this to 150%. The written report indicates that tea drinking communities are at an increased risk of dental and skeletal fluorosis, in the case where water fluoridation is in upshot.[27] Fluoride ion in low doses in the oral fissure reduces tooth decay.[28] For this reason, it is used in toothpaste and h2o fluoridation. At much college doses and frequent exposure, fluoride causes health complications and tin be toxic.
Applications [edit]
Fluoride salts and hydrofluoric acid are the main fluorides of industrial value. Compounds with C-F bonds fall into the realm of organofluorine chemical science. The chief uses of fluoride, in terms of volume, are in the production of cryolite, Na3AlF6. It is used in aluminium smelting. Formerly, it was mined, but now it is derived from hydrogen fluoride. Fluorite is used on a big scale to separate slag in steel-making. Mined fluorite (CaFtwo) is a commodity chemic used in steel-making.
Hydrofluoric acid and its anhydrous grade, hydrogen fluoride, is also used in the production of fluorocarbons. Hydrofluoric acrid has a diverseness of specialized applications, including its power to dissolve glass.[4]
Cavity prevention [edit]
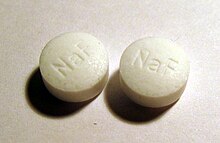
Fluoride is sold in tablets for cavity prevention.
Fluoride-containing compounds, such as sodium fluoride or sodium monofluorophosphate are used in topical and systemic fluoride therapy for preventing tooth decay. They are used for water fluoridation and in many products associated with oral hygiene.[29] Originally, sodium fluoride was used to fluoridate water; hexafluorosilicic acid (HtwoSiF6) and its salt sodium hexafluorosilicate (Na2SiF6) are more commonly used additives, peculiarly in the United States. The fluoridation of h2o is known to forestall tooth decay[30] [31] and is considered by the U.S. Centers for Disease Command and Prevention to be "one of 10 great public wellness achievements of the 20th century".[32] [33] In some countries where big, centralized water systems are uncommon, fluoride is delivered to the populace by fluoridating tabular array salt. For the method of action for crenel prevention, see Fluoride therapy. Fluoridation of water has its critics (run across Water fluoridation controversy).[34] Fluoridated toothpaste is in common employ. Meta-analysis show the efficacy of 500 ppm fluoride in toothpastes.[35] [36] Nevertheless, no beneficial upshot can be detected when more than 1 fluoride source is used for daily oral care.[37] [ demand quotation to verify ]
Biochemical reagent [edit]
Fluoride salts are commonly used in biological assay processing to inhibit the activeness of phosphatases, such as serine/threonine phosphatases.[38] Fluoride mimics the nucleophilic hydroxide ion in these enzymes' active sites.[39] Beryllium fluoride and aluminium fluoride are also used equally phosphatase inhibitors, since these compounds are structural mimics of the phosphate grouping and tin act as analogues of the transition land of the reaction.[40] [41]
Dietary recommendations [edit]
The U.Due south. Establish of Medicine (IOM) updated Estimated Average Requirements (EARs) and Recommended Dietary Allowances (RDAs) for some minerals in 1997. Where in that location was not sufficient information to establish EARs and RDAs, an judge designated Adequate Intake (AI) was used instead. AIs are typically matched to actual average consumption, with the assumption that in that location appears to be a demand, and that demand is met past what people consume. The current AI for women 19 years and older is iii.0 mg/day (includes pregnancy and lactation). The AI for men is 4.0 mg/twenty-four hours. The AI for children ages i–xviii increases from 0.7 to 3.0 mg/day. The major known risk of fluoride deficiency appears to be an increased risk of leaner-caused tooth cavities. As for safety, the IOM sets tolerable upper intake levels (ULs) for vitamins and minerals when show is sufficient. In the instance of fluoride the UL is ten mg/day. Collectively the EARs, RDAs, AIs and ULs are referred to equally Dietary Reference Intakes (DRIs).[42]
The European Nutrient Safe Authority (EFSA) refers to the collective set of information equally Dietary Reference Values, with Population Reference Intake (PRI) instead of RDA, and Average Requirement instead of EAR. AI and UL defined the same as in United States. For women ages 18 and older the AI is set at 2.9 mg/day (includes pregnancy and lactation). For men the value is 3.iv mg/day. For children ages one–17 years the AIs increase with age from 0.6 to three.2 mg/24-hour interval. These AIs are comparable to the U.S. AIs.[43] The EFSA reviewed safe evidence and prepare an adult UL at seven.0 mg/twenty-four hours (lower for children).[44]
For U.Due south. food and dietary supplement labeling purposes the amount of a vitamin or mineral in a serving is expressed every bit a percentage of Daily Value (%DV). Although there is information to set Acceptable Intake, fluoride does not have a Daily Value and is not required to be shown on food labels.[45]
Estimated daily intake [edit]
Daily intakes of fluoride can vary significantly according to the diverse sources of exposure. Values ranging from 0.46 to 3.6–5.4 mg/day have been reported in several studies (IPCS, 1984).[26] In areas where water is fluoridated this can be expected to be a significant source of fluoride, however fluoride is likewise naturally present in virtually all foods and beverages at a wide range of concentrations.[46] The maximum condom daily consumption of fluoride is 10 mg/twenty-four hours for an adult (U.S.) or 7 mg/day (European union).[42] [44]
The upper limit of fluoride intake from all sources (fluoridated water, food, beverages, fluoride dental products and dietary fluoride supplements) is set at 0.10 mg/kg/day for infants, toddlers, and children through to eight years old. For older children and adults, who are no longer at risk for dental fluorosis, the upper limit of fluoride is set at 10 mg/day regardless of weight.[47]
| Nutrient/Drink | Fluoride (mg per 1000g/ppm) | Portion | Fluoride (mg per portion) |
|---|---|---|---|
| Black tea (brewed) | iii.73 | ane cup, 240 g (viii fl oz) | 0.884 |
| Raisins, seedless | two.34 | small box, 43 yard (1.5 oz) | 0.101 |
| Table vino | 1.53 | Bottle, 750 ml (26.4 fl oz) | 1.150 |
| Municipal tap-water, (Fluoridated) | 0.81 | Recommended daily intake, iii litres (0.79 US gal) | two.433 |
| Broiled potatoes, Russet | 0.45 | Medium potato, 140 g (0.3 lb) | 0.078 |
| Lamb | 0.32 | Chop, 170 g (six oz) | 0.054 |
| Carrots | 0.03 | 1 large carrot, 72 g (two.5 oz) | 0.002 |
| Source: Data taken from United States Department of Agronomics, National Food Database Archived 2014-03-01 at the Wayback Machine[48] | |||
Safety [edit]
Ingestion [edit]
According to the U.S. Department of Agriculture, the Dietary Reference Intakes, which is the "highest level of daily nutrient intake that is probable to pose no chance of adverse wellness effects" specify 10 mg/day for about people, corresponding to 10 L of fluoridated water with no risk. For young children the values are smaller, ranging from 0.7 mg/d to two.ii mg/d for infants.[49] Water and food sources of fluoride include community h2o fluoridation, seafood, tea, and gelatin.[50]
Soluble fluoride salts, of which sodium fluoride is the most common, are toxic, and have resulted in both accidental and self-inflicted deaths from astute poisoning.[4] The lethal dose for most adult humans is estimated at 5 to 10 grand (which is equivalent to 32 to 64 mg elemental fluoride per kg body weight).[51] [52] [53] A instance of a fatal poisoning of an adult with iv grams of sodium fluoride is documented,[54] and a dose of 120 one thousand sodium fluoride has been survived.[55] For sodium fluorosilicate (Na2SiF6), the median lethal dose (LD50) orally in rats is 125 mg/kg, respective to 12.v g for a 100 kg adult.[56]
Treatment may involve oral administration of dilute calcium hydroxide or calcium chloride to forbid further assimilation, and injection of calcium gluconate to increase the calcium levels in the blood.[54] Hydrogen fluoride is more unsafe than salts such every bit NaF because it is corrosive and volatile, and tin can result in fatal exposure through inhalation or upon contact with the skin; calcium gluconate gel is the usual antidote.[57]
In the higher doses used to treat osteoporosis, sodium fluoride can cause hurting in the legs and incomplete stress fractures when the doses are as well loftier; it also irritates the tum, sometimes and then severely equally to cause ulcers. Slow-release and enteric-coated versions of sodium fluoride do not have gastric side furnishings in whatever meaning fashion, and have milder and less frequent complications in the basic.[58] In the lower doses used for water fluoridation, the only articulate agin effect is dental fluorosis, which can alter the appearance of children's teeth during tooth evolution; this is more often than not balmy and is unlikely to stand for any real effect on aesthetic appearance or on public wellness.[59] Fluoride was known to raise the measurement of bone mineral density at the lumbar spine, but it was not constructive for vertebral fractures and provoked more not vertebral fractures.[60]
A popular urban myth claims that the Nazis used fluoride in concentration camps, but there is no historical testify to prove this claim.[61]
In areas that have naturally occurring high levels of fluoride in groundwater which is used for drinking water, both dental and skeletal fluorosis can exist prevalent and astringent.[62]
Hazard maps for fluoride in groundwater [edit]
Around one-third of the human population drinks h2o from groundwater resources. Of this, about 10%, approximately three hundred meg people, obtains water from groundwater resources that are heavily contaminated with arsenic or fluoride.[63] These trace elements derive mainly from minerals.[64] Maps locating potential problematic wells are available.[65]
Topical [edit]
Concentrated fluoride solutions are corrosive.[66] Gloves made of nitrile rubber are worn when handling fluoride compounds. The hazards of solutions of fluoride salts depend on the concentration. In the presence of stiff acids, fluoride salts release hydrogen fluoride, which is corrosive, especially toward glass.[four]
Other derivatives [edit]
Organic and inorganic anions are produced from fluoride, including:
- Bifluoride, used as an etchant for drinking glass[67]
- Tetrafluoroberyllate
- Hexafluoroplatinate
- Tetrafluoroborate used in organometallic synthesis
- Hexafluorophosphate used as an electrolyte in commercial secondary batteries.
- Trifluoromethanesulfonate
See also [edit]
- Fluorine-19 nuclear magnetic resonance spectroscopy
- Fluoride deficiency
- Fluoride selective electrode
- Fluoride therapy
- Sodium monofluorophosphate
References [edit]
- ^ "Fluorides – PubChem Public Chemic Database". The PubChem Project. USA: National Center for Biotechnology Data. Identification.
- ^ Chase, Chiliad. Due west. (1998). "Fluorine anion". NIST: 1–1951. Retrieved iv July 2012.
- ^ Wells, J.C. (2008). Longman pronunciation dictionary (3rd ed.). Harlow, England: Pearson Teaching Limited/Longman. p. 313. ISBN9781405881180. . Co-ordinate to this source, is a possible pronunciation in British English.
- ^ a b c d Aigueperse, Jean; Mollard, Paul; Devilliers, Didier; Chemla, Marius; Faron, Robert; Romano, René; Cuer, Jean Pierre (2000). "Fluorine Compounds, Inorganic". Ullmann's Encyclopedia of Industrial Chemical science. doi:10.1002/14356007.a11_307. ISBN978-3527306732.
- ^ "Public Health Statement for Fluorides, Hydrogen Fluoride, and Fluorine". ATSDR. September 2003.
- ^ "Ambience Water Quality Criteria for Fluoride". Government of British Columbia. Archived from the original on 24 September 2015. Retrieved viii October 2014.
- ^ Liteplo, Dr R.; Gomes, R.; Howe, P.; Malcolm, Heath (2002). FLUORIDES - Environmental Health Criteria 227 : 1st typhoon. Geneva: Globe Health Organisation. ISBN978-9241572279.
- ^ a b Fawell, J.Thousand.; et al. "Fluoride in Drinking-h2o Background document for development of WHO Guidelines for Drinking-h2o Quality" (PDF). World Health Organization. Retrieved 6 May 2016.
- ^ Yadav, Krishna Kumar; Kumar, Sandeep; Pham, Quoc Bao; Gupta, Neha; Rezania, Shahabaldin; Kamyab, Hesam; Yadav, Shalini; Vymazal, January; Kumar, Vinit; Tri, Doan Quang; Talaiekhozani, Amirreza; Prasad, Shiv; Reece, Lisa M.; Singh, Neeraja; Maurya, Pradip Kumar; Cho, Jinwoo (Oct 2019). "Fluoride contamination, health issues and remediation methods in Asian groundwater: A comprehensive review". Ecotoxicology and Ecology Safety. 182: 109362. doi:ten.1016/j.ecoenv.2019.06.045. PMID 31254856. S2CID 195764865.
- ^ Tiemann, Mary (5 April 2013). "Fluoride in Drinking H2o: A Review of Fluoridation and Regulation Issues" (PDF). Congressional Inquiry Service. p. iii. Retrieved vi May 2016.
- ^ Chandio, Tasawar Ali; Khan, Muhammad Nasiruddin; Muhammad, Maria Taj; Yalcinkaya, Ozcan; Wasim, Agha Arslan; Kayis, Ahmet Furkan (Jan 2021). "Fluoride and arsenic contamination in drinking water due to mining activities and its impact on local area population". Environmental Scientific discipline and Pollution Research. 28 (2): 2355–2368. doi:ten.1007/s11356-020-10575-ix. PMID 32880840. S2CID 221463681.
- ^ Bellomo, Sergio; Aiuppa, Alessandro; D'Alessandro, Walter; Parello, Francesco (Baronial 2007). "Environmental impact of magmatic fluorine emission in the Mt. Etna area". Periodical of Volcanology and Geothermal Research. 165 (1–two): 87–101. Bibcode:2007JVGR..165...87B. doi:10.1016/j.jvolgeores.2007.04.013.
- ^ Smith, Frank A.; Hodge, Harold C.; Dinman, B. D. (9 Jan 2009). "Airborne fluorides and homo: Part I". CRC Critical Reviews in Environmental Command. 8 (1–four): 293–371. doi:10.1080/10643387709381665.
- ^ Smith, Frank A.; Hodge, Harold C.; Dinman, B. D. (9 January 2009). "Airborne fluorides and human: Part Two". CRC Critical Reviews in Ecology Control. 9 (i): i–25. doi:10.1080/10643387909381666.
- ^ Arnesen, A.K.Thousand.; Abrahamsen, G.; Sandvik, G.; Krogstad, T. (February 1995). "Aluminium-smelters and fluoride pollution of soil and soil solution in Kingdom of norway". Science of the Full Environment. 163 (1–3): 39–53. Bibcode:1995ScTEn.163...39A. doi:10.1016/0048-9697(95)04479-K.
- ^ Wong MH, Fung KF, Carr HP (2003). "Aluminium and fluoride contents of tea, with emphasis on brick tea and their health implications". Toxicology Letters. 137 (1–2): 111–20. doi:10.1016/S0378-4274(02)00385-5. PMID 12505437.
- ^ Malinowska East, Inkielewicz I, Czarnowski W, Szefer P (2008). "Cess of fluoride concentration and daily intake by human from tea and herbal infusions". Food Chem. Toxicol. 46 (3): 1055–61. doi:10.1016/j.fct.2007.x.039. PMID 18078704.
- ^ Gardner EJ, Ruxton CH, Leeds AR (2007). "Blackness tea--helpful or harmful? A review of the testify". European Journal of Clinical Diet. 61 (i): 3–xviii. doi:10.1038/sj.ejcn.1602489. PMID 16855537.
- ^ Wiberg; Holleman, A.F. (2001). Inorganic chemistry (1st English ed., [edited] by Nils Wiberg. ed.). San Diego, Calif. : Berlin: Academic Press, W. de Gruyter. ISBN978-0-12-352651-9.
- ^ Schwesinger, Reinhard; Link, Reinhard; Wenzl, Peter; Kossek, Sebastian (2005). "Anhydrous Phosphazenium Fluorides as Sources for Extremely Reactive Fluoride Ions in Solution". Chemistry. 12 (2): 438–45. doi:10.1002/chem.200500838. PMID 16196062.
- ^ Haoran Sun & Stephen M. DiMagno (2005). "Anhydrous Tetrabutylammonium Fluoride". Journal of the American Chemical Society. 127 (7): 2050–1. doi:10.1021/ja0440497. PMID 15713075.
- ^ Bennett, Brian K.; Harrison, Roger G.; Richmond, Thomas G. (1994). "Cobaltocenium Fluoride: A Novel Source of "Naked" Fluoride Formed by Carbon-Fluorine Bond Activation in a Saturated Perfluorocarbon". Journal of the American Chemical Guild. 116 (24): 11165–11166. doi:10.1021/ja00103a045.
- ^ Alič, B.; Tavčar, G. (2016). "Reaction of N-heterocyclic carbene (NHC) with different HF sources and ratios – A free fluoride reagent based on imidazolium fluoride". J. Fluorine Chem. 192: 141–146. doi:10.1016/j.jfluchem.2016.11.004.
- ^ Alič, B.; Tramšek, M.; Kokalj, A.; Tavčar, G. (2017). "Detached GeF5– Anion Structurally Characterized with a Readily Synthesized Imidazolium Based Naked Fluoride Reagent". Inorg. Chem. 56 (16): 10070–10077. doi:ten.1021/acs.inorgchem.7b01606. PMID 28792216.
- ^ Zupanek, Ž.; Tramšek, M.; Kokalj, A.; Tavčar, G. (2018). "Reactivity of VOF3 with N-Heterocyclic Carbene and Imidazolium Fluoride: Analysis of Ligand–VOF3 Bonding with Evidence of a Minute π Back-Donation of Fluoride". Inorg. Chem. 57 (21): 13866–13879. doi:x.1021/acs.inorgchem.8b02377. PMID 30353729. S2CID 53031199.
- ^ a b Fawell, J. "Fluoride in Drinking-water" (PDF). Globe Wellness Organization. Retrieved 10 March 2016.
- ^ a b c Chan, Laura; Mehra, Aradhana; Saikat, Sohel; Lynch, Paul (May 2013). "Human exposure assessment of fluoride from tea (Camellia sinensis 50.): A UK based issue?". Nutrient Research International. 51 (2): 564–570. doi:x.1016/j.foodres.2013.01.025.
- ^ "Fluoride Free Toothpaste – Fluoride (Finally!) Explained". 27 June 2016.
- ^ McDonagh Yard. S.; Whiting P. F.; Wilson P. Grand.; Sutton A. J.; Chestnutt I.; Cooper J.; Misso K.; Bradley M.; Treasure Eastward.; Kleijnen J. (2000). "Systematic review of water fluoridation". British Medical Journal. 321 (7265): 855–859. doi:ten.1136/bmj.321.7265.855. PMC27492. PMID 11021861.
- ^ Griffin And so, Regnier Eastward, Griffin PM, Huntley Five (2007). "Effectiveness of fluoride in preventing caries in adults". J. Paring. Res. 86 (5): 410–five. doi:10.1177/154405910708600504. hdl:10945/60693. PMID 17452559. S2CID 58958881.
- ^ Winston A. E.; Bhaskar South. N. (1 November 1998). "Caries prevention in the 21st century". J. Am. Dent. Assoc. 129 (eleven): 1579–87. doi:10.14219/jada.archive.1998.0104. PMID 9818575. Archived from the original on fifteen July 2012.
- ^ "Community Water Fluoridation". Centers for Affliction Control and Prevention. Retrieved x March 2016.
- ^ "X Peachy Public Health Achievements in the 20th Century". Centers for Disease Control and Prevention. Archived from the original on 13 March 2016. Retrieved x March 2016.
- ^ Newbrun E (1996). "The fluoridation war: a scientific dispute or a religious argument?". Periodical of Public Wellness Dentistry. 56 (5 Spec No): 246–52. doi:10.1111/j.1752-7325.1996.tb02447.ten. PMID 9034969.
- ^ Walsh, Tanya; Worthington, Helen Five.; Glenny, Anne-Marie; Marinho, Valeria Cc; Jeroncic, Ana (iv March 2019). "Fluoride toothpastes of different concentrations for preventing dental caries". Cochrane Database of Systematic Reviews. 3 (3): CD007868. doi:x.1002/14651858.CD007868.pub3. ISSN 1469-493X. PMC6398117. PMID 30829399.
- ^ "Remineralization of initial carious lesions in deciduous enamel afterwards application of dentifrices of different fluoride concentrations". springermedizin.de (in German). Retrieved 24 February 2021.
- ^ Hausen, H.; Kärkkäinen, Southward.; Seppä, L. (February 2000). "Application of the loftier-chance strategy to command dental caries". Community Dentistry and Oral Epidemiology. 28 (1): 26–34. doi:10.1034/j.1600-0528.2000.280104.x. ISSN 0301-5661. PMID 10634681.
- ^ Nakai C, Thomas JA (1974). "Backdrop of a phosphoprotein phosphatase from bovine middle with activity on glycogen synthase, phosphorylase, and histone". J. Biol. Chem. 249 (20): 6459–67. doi:x.1016/S0021-9258(nineteen)42179-0. PMID 4370977.
- ^ Schenk G, Elliott TW, Leung E, et al. (2008). "Crystal structures of a regal acrid phosphatase, representing dissimilar steps of this enzyme's catalytic cycle". BMC Struct. Biol. 8: half-dozen. doi:ten.1186/1472-6807-8-six. PMC2267794. PMID 18234116.
- ^ Wang Due west, Cho HS, Kim R, et al. (2002). "Structural characterization of the reaction pathway in phosphoserine phosphatase: crystallographic "snapshots" of intermediate states". J. Mol. Biol. 319 (ii): 421–31. doi:10.1016/S0022-2836(02)00324-8. PMID 12051918.
- ^ Cho H, Wang W, Kim R, et al. (2001). "BeF(iii)(-) acts as a phosphate analog in proteins phosphorylated on aspartate: structure of a BeF(iii)(-) circuitous with phosphoserine phosphatase". Proc. Natl. Acad. Sci. U.s.a.A. 98 (fifteen): 8525–thirty. Bibcode:2001PNAS...98.8525C. doi:10.1073/pnas.131213698. PMC37469. PMID 11438683.
- ^ a b Institute of Medicine (1997). "Fluoride". Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D and Fluoride. Washington, DC: The National Academies Press. pp. 288–313. doi:10.17226/5776. ISBN978-0-309-06403-three. PMID 23115811.
- ^ "Overview on Dietary Reference Values for the EU population as derived by the EFSA Panel on Dietetic Products, Nutrition and Allergies" (PDF). 2017.
- ^ a b Tolerable Upper Intake Levels For Vitamins And Minerals (PDF), European Food Safety Authority, 2006
- ^ "Federal Register May 27, 2016 Food Labeling: Revision of the Nutrition and Supplement Facts Labels. FR page 33982" (PDF).
- ^ "Nutrient Lists". Agricultural Enquiry Service Usa Department of Agriculture. Archived from the original on 26 May 2014. Retrieved 25 May 2014.
- ^ Levy, Steven Chiliad.; Guha-Chowdhury, Nupur (1999). "Total Fluoride Intake and Implications for Dietary Fluoride Supplementation". Journal of Public Wellness Dentistry. 59 (iv): 211–223. doi:10.1111/j.1752-7325.1999.tb03272.x. PMID 10682326.
- ^ "Food Composition Databases: Food Search: Fluoride". Agronomical Research Service, United States Section of Agriculture. Archived from the original on 5 December 2018. Retrieved 5 December 2018.
- ^ "Dietary Reference Intakes: EAR, RDA, AI, Acceptable Macronutrient Distribution Ranges, and UL". Usa Department of Agriculture. Retrieved 9 September 2017.
- ^ "Fluoride in diet". U.S. National Library of Medicine. Retrieved 10 March 2016.
- ^ Gosselin, RE; Smith RP; Hodge HC (1984). Clinical toxicology of commercial products. Baltimore (Physician): Williams & Wilkins. pp. III–185–93. ISBN978-0-683-03632-9.
- ^ Baselt, RC (2008). Disposition of toxic drugs and chemicals in homo. Foster City (CA): Biomedical Publications. pp. 636–40. ISBN978-0-9626523-seven-0.
- ^ IPCS (2002). Environmental health criteria 227 (Fluoride). Geneva: International Programme on Chemical Condom, World Health System. p. 100. ISBN978-92-four-157227-9.
- ^ a b Rabinowitch, IM (1945). "Astute Fluoride Poisoning". Canadian Medical Association Journal. 52 (4): 345–9. PMC1581810. PMID 20323400.
- ^ Abukurah AR, Moser AM Jr, Baird CL, Randall RE Jr, Setter JG, Blanke RV (1972). "Acute sodium fluoride poisoning". JAMA. 222 (7): 816–vii. doi:10.1001/jama.1972.03210070046014. PMID 4677934.
- ^ The Merck Index, 12th edition, Merck & Co., Inc., 1996
- ^ Muriale Fifty, Lee Due east, Genovese J, Tendency S (1996). "Fatality due to acute fluoride poisoning following dermal contact with hydrofluoric acrid in a palynology laboratory". Ann. Occup. Hyg. twoscore (half dozen): 705–710. doi:ten.1016/S0003-4878(96)00010-5. PMID 8958774.
- ^ Murray TM, Ste-Marie LG (1996). "Prevention and management of osteoporosis: consensus statements from the Scientific Advisory Board of the Osteoporosis Society of Canada. 7. Fluoride therapy for osteoporosis". CMAJ. 155 (vii): 949–54. PMC1335460. PMID 8837545.
- ^ National Health and Medical Research Council (Commonwealth of australia) (2007). A systematic review of the efficacy and condom of fluoridation (PDF). ISBN978-i-86496-415-eight. Archived from the original (PDF) on xiv October 2009. Retrieved 21 February 2010. Summary: Yeung CA (2008). "A systematic review of the efficacy and safety of fluoridation". Evid.-Based Paring. 9 (2): 39–43. doi:10.1038/sj.ebd.6400578. PMID 18584000.
- ^ Haguenauer, D; Welch, Five; Shea, B; Tugwell, P; Adachi, JD; Wells, 1000 (2000). "Fluoride for the treatment of postmenopausal osteoporotic fractures: a meta-analysis". Osteoporosis International. 11 (9): 727–38. doi:10.1007/s001980070051. PMID 11148800. S2CID 538666.
- ^ Bowers, Becky (six October 2011). "Truth nearly fluoride doesn't include Nazi myth". PolitiFact.com. Tampa Bay Times. Retrieved 26 March 2015.
- ^ World Health Organization (2004). "Fluoride in drinking-water" (PDF). Archived from the original (PDF) on 4 March 2016. Retrieved thirteen February 2014.
- ^ Eawag (2015) Geogenic Contagion Handbook – Addressing Arsenic and Fluoride in Drinking Water. C.A. Johnson, A. Bretzler (Eds.), Swiss Federal Institute of Aquatic Science and Engineering (Eawag), Duebendorf, Switzerland. (download: www.eawag.ch/en/inquiry/humanwelfare/drinkingwater/wrq/geogenic-contamination-handbook/)
- ^ Rodríguez-Lado, L.; Sun, G.; Berg, M.; Zhang, Q.; Xue, H.; Zheng, Q.; Johnson, C.A. (2013). "Groundwater arsenic contamination throughout Red china". Scientific discipline. 341 (6148): 866–868. Bibcode:2013Sci...341..866R. doi:x.1126/science.1237484. PMID 23970694. S2CID 206548777.
- ^ Groundwater Assessment Platform
- ^ Nakagawa Thou, Matsuya S, Shiraishi T, Ohta Grand (1999). "Outcome of fluoride concentration and pH on corrosion beliefs of titanium for dental apply". Periodical of Dental Research. 78 (9): 1568–72. doi:10.1177/00220345990780091201. PMID 10512392. S2CID 32650790.
- ^ "Ammonium bifluoride in the glass manufacture - Chimex Ltd".
External links [edit]
![]()
Wikimedia Commons has media related to Fluorides.
- "Fluoride in Drinking H2o: A Review of Fluoridation and Regulation Bug", Congressional Research Service
- U.S. authorities site for checking status of local water fluoridation
Chemical Formula For Aluminum Fluoride,
Source: https://en.wikipedia.org/wiki/Fluoride
Posted by: berniertheirm.blogspot.com

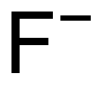



0 Response to "Chemical Formula For Aluminum Fluoride"
Post a Comment